Managing clinical trials is no easy task, with a range of challenges that can impact the success of a study. To gain deeper insights into these hurdles, we recently asked industry professionals to identify their biggest challenge in clinical trial management.
Poll Overview: What Are the Biggest Challenges?
We asked industry professionals: What’s the most significant challenge you face in clinical trial management? The options provided were:
- Robust data quality
- More complex trial designs
- Accurate patient forecasting
- Integration of technology
Each of these options reflects a critical aspect of trial management that can influence the success of clinical research. Below we provide a closer look at the poll results and what they mean for the industry.
Key Poll Results
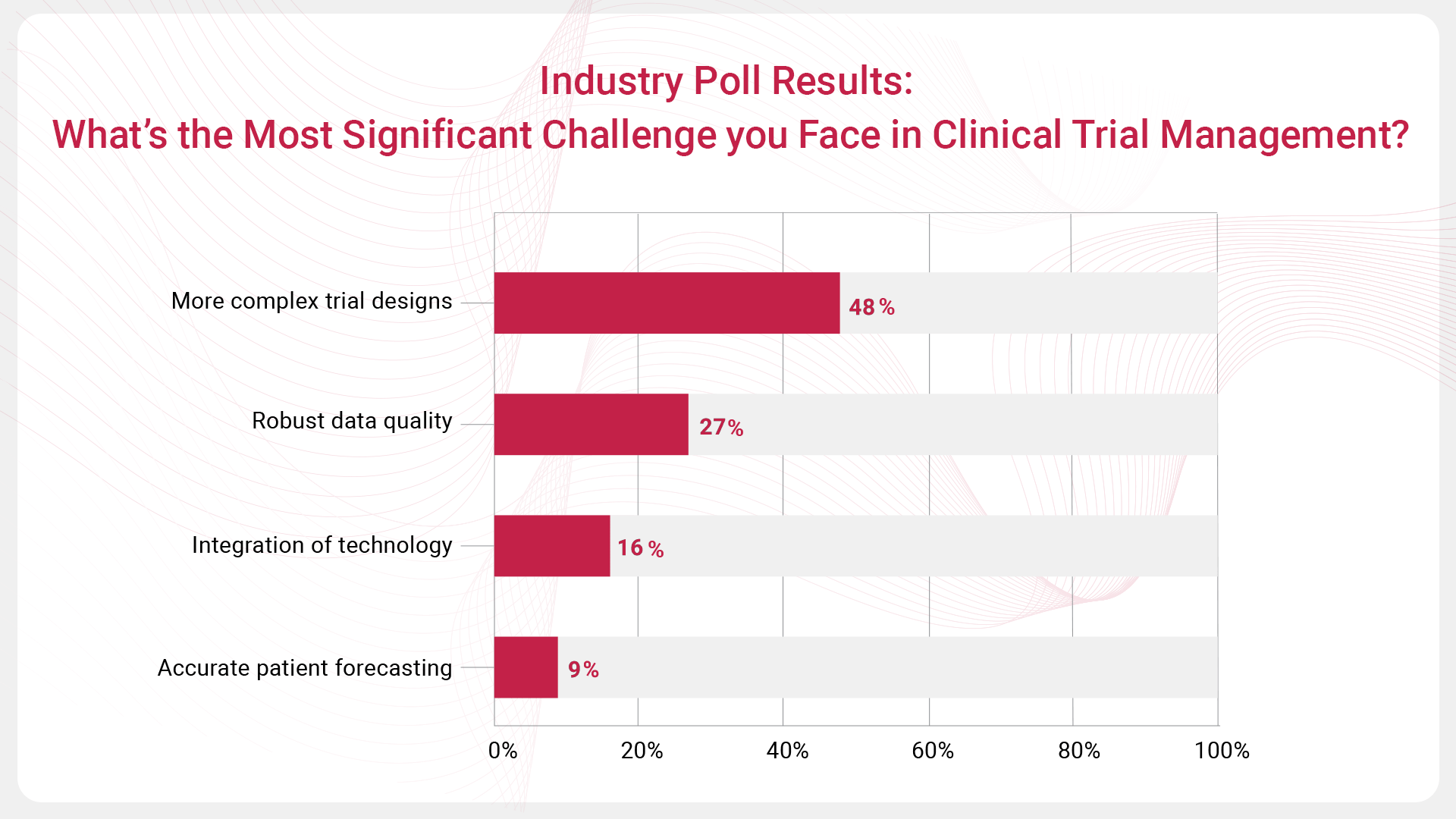
- More complex trial designs emerged as the top challenge, with 48% of respondents selecting this option. As clinical trials increasingly involve sophisticated methodologies such as adaptive designs, managing these complexities requires expertise, advanced planning and stakeholder coordination. The growing complexity of trial designs underscores the need for innovative strategies and tools to handle the intricacies of modern clinical research.
- Robust data quality was the second most cited challenge, with 27% of the vote. Ensuring high-quality, reliable data is essential for drawing valid conclusions about the safety and efficacy of new treatments. Maintaining data integrity across diverse sources and formats is a significant concern for trial managers, highlighting the importance of strong data management capabilties and quality assurance processes.
- Integration of technology was identified by 16% of respondents as a significant challenge. With the rise of digital tools, wearables, and electronic health records, the integration of technology into clinical trials is both an opportunity and a challenge. Successfully incorporating these technologies requires careful planning and a clear understanding of how they can best be used to enhance trial efficiency and data collection.
- Accurate patient forecasting was selected by 9% of respondents, indicating that while this is a critical factor, it is perceived as less challenging than the other options. Accurate patient forecasting is vital for maintaining timelines and ensuring sufficient enrolment.
What These Results Mean for Clinical Trial Management
The poll results clearly highlight the increasing complexity of clinical trial designs as the most pressing challenge. This trend points to a need for more sophisticated project management approaches, better training, and enhanced collaboration among stakeholders. Additionally, the emphasis on robust data quality and the integration of technology, along with accelerating drug development—an essential factor in the industry—suggests that these areas will continue to be central to ongoing innovation and advancement.
For organizations managing clinical trials, these insights emphasize the importance of investing in the right tools, technologies, and expertise to navigate these challenges effectively. As the landscape of clinical trials continues to evolve, those who can adapt to these complexities will be better positioned to succeed.
Conclusion
Clinical trial management has many potential challenges, from increasingly complex designs to ensuring robust data quality and effectively integrating new technologies. Our poll results provide valuable insights into where the industry is focused and what challenges lie ahead. By understanding these key areas, we can better support our clients in overcoming these hurdles and achieving successful trial outcomes.
Ready to overcome the challenges of clinical trial management? If you’re looking to turn these challenges into opportunities for success, we’re here to help.



